The Importance of Strain
Within each species of bacteria there is a multitude of strains. Some strains within a given species are strong and resilient, with a demonstrated capacity to survive the trip through the upper GIT, interact with the immune system, and inhibit pathogenic bacteria, whereas other strains are weak and cannot even survive transit through the stomach or small intestine. This is an extremely important concept to know, as it means that just because one strain of bacteria in a given species has a proven action or characteristic, it does not mean that another strain will too, even if they are closely related.
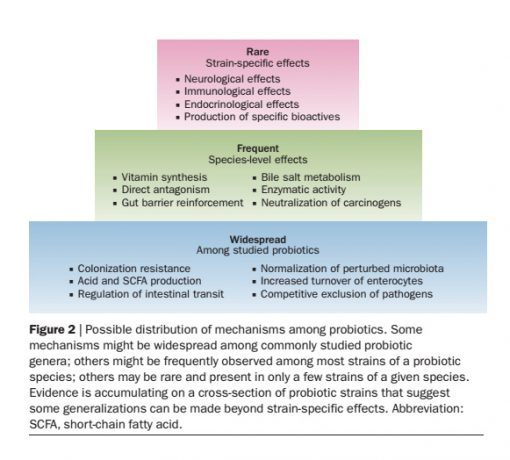
Strains of bacteria within the same species can have significantly different actions, properties and characteristics, as many of these characteristics are strain-specific qualities.[1, 2] As an example, Lactobacillus plantarum strain 299v has been shown to effectively reduce IBS symptoms in a number of trials [3, 4], whereas administration of L. plantarum strain MF1298 was found to actually worsen IBS symptoms.[5] To achieve desired therapeutic outcomes, it is vital to prescribe the specific probiotic strains that have demonstrated the therapeutic qualities required and clinical efficacy in the condition in question (when possible), as strains of bacteria, even those within the same species, can have significantly different therapeutic actions, properties and characteristics.
It should also be noted that a probiotic strain that works in one condition will not necessarily be effective for other conditions. For example, the multistrain preparation commonly referred to as Lab4 (containing Lactobacillus acidophilus strains CUL21 and CUL60, Bifidobacterium bifidum CUL20 and Bifidobacterium lactis CUL34) appears effective in the management of IBS,[6] but is ineffective in the prevention of antibiotic-associated side effects.[7] Another example is the capacity of Lactobacillus rhamnosus GG to effectively prevent antibiotic-associated side effects,[8] but not urinary tract infections.[9]
Unfortunately, this concept of strain specificity is not well known amongst clinicians or consumers of probiotics, leading to a vulnerability to misunderstandings and often inaccurate extrapolations from the research literature. For example, some supplement manufacturers will quote a study that utilized L. rhamnosus strain HN001 and then say that their probiotic supplement containing a strain of L. fermentum or another strain of L. rhamnosus will do the same. This is quite incorrect. Unless proven, one cannot assume that a given strain of L. rhamnosus, L. acidophilus, B. bifidum or any other species of lactic acid bacteria will survive transit through the upper GIT, let alone have specific therapeutic actions. Again reiterating why best practice is to prescribe the probiotic strain(s) that have proven therapeutic efficacy in the condition being treated when possible.[10]
Are all attributes and actions strain specific?
Obviously there is enough similarity between strains in a given species that they are still considered members of the same species. There is, however, significant evidence showing that even basic attributes can differ between strains.[2] Unfortunately, without detailed comparative studies, it is impossible to know how much two strains within a given species differ, in what ways they differ, and, most importantly, whether these differences will impact therapeutic effectiveness. Research has shown that these differences can determine whether a strain is effective or not.[3, 5, 11]
For some indications, differences between strains may be of little consequence. For example, strains of Lactobacillus delbrueckii subsp. bulgaricus appear to almost universally have the capacity to produce lactase. It is generally agreed that any strain within this species will aid in lactose digestion and, hence, would be of benefit in the management of lactose intolerance (if taken concurrently with a lactose load). Other indications where strain specificity may be of less importance include normalization of a perturbed microbiota and acidification of the GIT via the production of lactic- and other short chain fatty acids.[12]
When treating specific disease conditions, however, due to the potential for substantial differences between strains, it is recommended to utilize those strains with demonstrated efficacy in that condition.
1. Guarner, F., et al., Probiotics and prebiotics. World Gastroenterology Organisation Global Guideline, 2011.
2. Marteau, P., Evidence of probiotic strain specificity makes extrapolation of results impossible from a strain to another, even from the same species. Annals of Gastroenterology & Hepatology, 2011. 2(1): p. 34-36.
3. Ducrotte, P., P. Sawant, and V. Jayanthi, Clinical trial: Lactobacillus plantarum 299v (DSM 9843) improves symptoms of irritable bowel syndrome. World J Gastroenterol, 2012. 18(30): p. 4012-8.
4. Niedzielin, K., H. Kordecki, and B. Birkenfeld, A controlled, double-blind, randomized study on the efficacy of Lactobacillus plantarum 299V in patients with irritable bowel syndrome. European Journal of Gastroenterology and Hepatology, 2001. 13: p. 1143-1147.
5. Ligaarden, S.C., et al., A candidate probiotic with unfavourable effects in subjects with irritable bowel syndrome: a randomised controlled trial. BMC Gastroenterol, 2010. 10: p. 16.
6. Williams, E.A., et al., Clinical trial: a multistrain probiotic preparation significantly reduces symptoms of irritable bowel syndrome in a double-blind placebo-controlled study. Aliment Pharmacol Ther, 2008. 29: p. 97-103.
7. Allen, S.J., et al., Lactobacilli and bifidobacteria in the prevention of antibiotic-associated diarrhoea and Clostridium difficile diarrhoea in older inpatients (PLACIDE): a randomised, double-blind, placebo-controlled, multicentre trial. The Lancet, 2013. 382(9900): p. 1249-1257.
8. Videlock, E.J. and F. Cremonini, Meta-analysis: probiotics in antibiotic-associated diarrhoea. Alimentary Pharmacology & Therapeutics, 2012. 35(12): p. 1355-1369.
9. Kontiokari, T., et al., Randomised trial of cranberry-lingonberry juice and Lactobacillus GG drink for the prevention of urinary tract infections in women. British Medical Journal, 2001. 322: p. 1571.
10. Hawrelak, J.A., Probiotics, in Textbook of Natural Medicine, J. Pizzorno and M. Murray, Editors. 2013, Elsevier: St Louis. p. 979-994.
11. Majamaa, H., et al., Lactic acid bacteria in the treatment of acute rotavirus gastroenteritis. Journal of Pediatric Gastroenterology and Nutrition, 1995. 20: p. 333-338.
12. Hill, C., et al., Expert consensus document: The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol, 2014. 11(8): p. 506-514.
